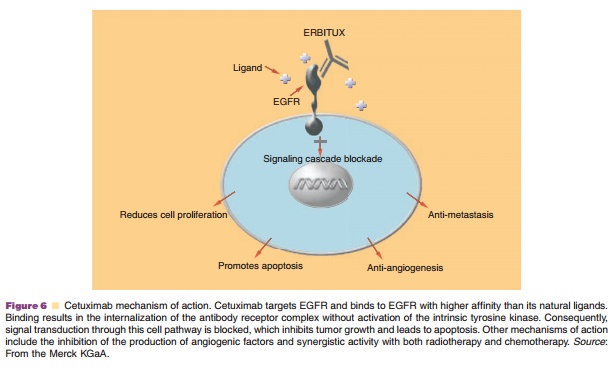
File:Cetuximab-Reconstitutes-Pro-Inflammatory-Cytokine-Secretions-and-Tumor-Infiltrating-Capabilities-of-video 1.ogv - Wikimedia Commons

Chemioterapici: Lista Di Anticorpi Monoclonali Approvati Per USO Clinico, Terapia Con Anticorpi Monoclonali, Denosumab, Cetuximab, Ipilimumab - Fonte Wikipedia: 9781232638650 - AbeBooks

Cetuximab-induced anaphylaxis and IgE specific for galactose-alpha-1,3-galactose. - Abstract - Europe PMC

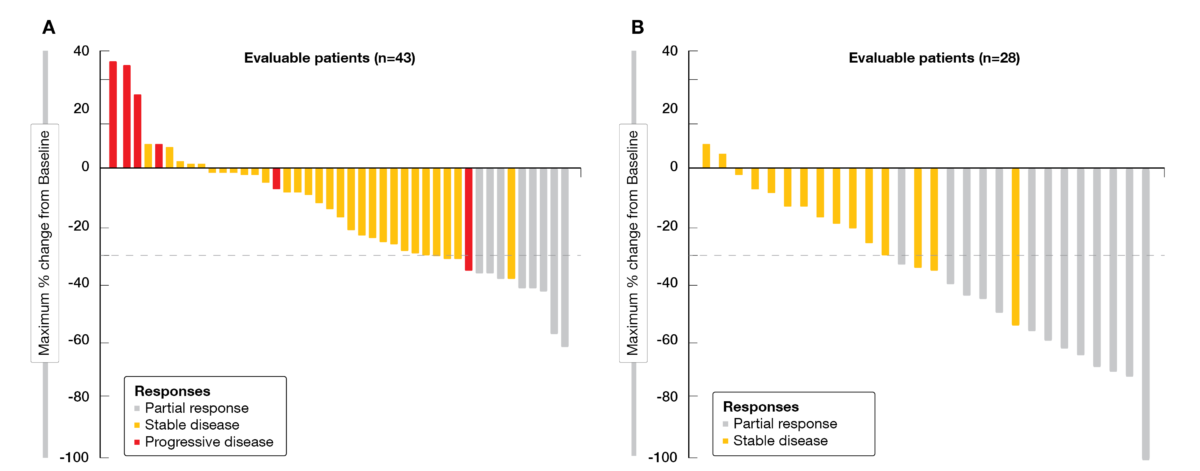
How Standard Is Second-Line Cetuximab in Recurrent or Metastatic Head and Neck Cancer in 2017? | Journal of Clinical Oncology

Molecular dissection of colorectal cancer in pre-clinical models identifies biomarkers predicting sensitivity to EGFR inhibitors | Nature Communications

Emergence of KRAS mutations and acquired resistance to anti-EGFR therapy in colorectal cancer. - Abstract - Europe PMC

PDF) RAS testing and cetuximab treatment for metastatic colorectal cancer: A cost-effectiveness analysis in a setting with limited health resources

Activation of ERBB2 Signaling Causes Resistance to the EGFR-Directed Therapeutic Antibody Cetuximab | Science Translational Medicine









.png)